




1 / 5
 Medovia Medical
Medovia Medical













| Technical Info | Specifications |
|---|---|
| Machine Type | Ethylene Oxide sterilizers / Sterilization chambers |
| Chamber Capacity | 1 to 100 m3 |
| Chamber Type | Rectangular, 1 to 13 pallets (80x120cm) |
| Doors Type | Double doors with pneumatic sliding (Manual/Auto) |
| Chamber Material | Stainless steel 304 or 316 |
| Hot Water Circulation | Double jacket with 4 circulation systems |
| Electric Energy | 380V AC, 50Hz or 60Hz |
| Software / Controls | |
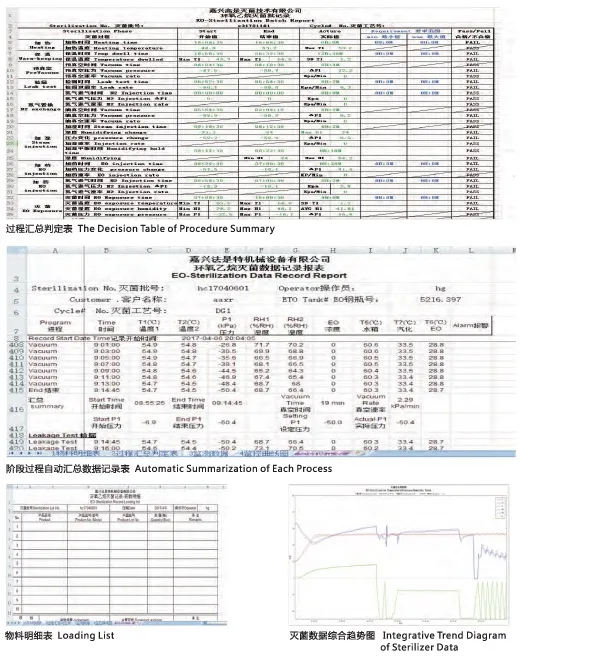
| Control System | Automatic/Manual control, Graphic recorder, Safety password |
| PLC | SIEMENS or SCHNEIDER |
| Sensors | Pressure, Temperature, Humidity sensors |
| Options & Systems | |
| Nitrogen Generator | Available |
| Software Compliance | FDA 21CFR part 11 qualified |


• Drafting technical agreements
• Custom manufacturing plans
• Equipment design
• Installation & operation training
• Overseas engineering service
• Maintenance & parts supply